


Announcing: EHC Emergency Displacement Fund (EDF)
With enduring shock and sadness, we think of our community members and others affected by the current challenging times, and are working tirelessly to provide support where possible. The EHC have been working with...
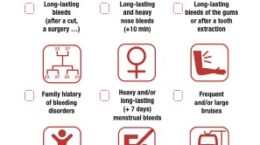
EHC Women’s Committee marks International Women’s Day

EHC VWD Working Group holds a strategic retreat

Meet Daria Mironova, EHC Communications and Public Policy Officer

BioMarin reports a serious adverse event for its phase I/II gene therapy trial for haemophilia A
February 4, 2022 – BioMarin’s gene therapy (valoctocogene roxaparvovec) for haemophilia A is currently in development and not yet licensed by regulators. BioMarin has completed dosing 134 patients in its phase 3 clinical study...

FDA places the Pfizer/Sangamo Therapeutics phase 3 AFFINE haemophilia A gene therapy study on clinical hold

EHCucate goes live






 Jim is aged 66 and has Haemophilia A with a high titer inhibitor which he developed at age 14.
Jim is aged 66 and has Haemophilia A with a high titer inhibitor which he developed at age 14. Maria Elisa Mancuso (MD, PhD) is a Haematologist and works as a Senior Haematology Consultant at the Center for Thrombosis and Haemorrhagic Diseases of IRCCS Humanitas Research Hospital in Rozzano, Milan, Italy. She is Adjunct Clinical Professor at Humanitas University. She obtained a post-degree in Clinical and Experimental Haematology and a PhD in Clinical Methodology. She is involved in clinical research and has published several original articles in peer-reviewed journals a The Lancet, Blood, Journal of Thrombosis and Haemostasis, Haematologica, Thrombosis and Haemostasis, British Journal of Haematology and Haemophilia. She is reviewer for several peer-reviewed journals and member of the Editorial Board of JTH. She is a member of several scientific societies (ISTH, WFH, ASH, EAHAD, SISET, AICE) and was a medical member of the Inhibitor Working Group of the European Hemophilia Consortium. She is co-chair of the ADVANCE Study Group. She has acted also as co-chair of the Scientific and Standardization Subcommittee of ISTH on FVIII, FIX and rare bleeding disorders. She has been involved as principal and co-investigator in several clinical trials, and she takes care of both children and adults with hemophilia and other congenital bleeding disorders with a specific scientific interest in novel therapies, prophylaxis, inhibitors, and chronic hepatitis C.
Maria Elisa Mancuso (MD, PhD) is a Haematologist and works as a Senior Haematology Consultant at the Center for Thrombosis and Haemorrhagic Diseases of IRCCS Humanitas Research Hospital in Rozzano, Milan, Italy. She is Adjunct Clinical Professor at Humanitas University. She obtained a post-degree in Clinical and Experimental Haematology and a PhD in Clinical Methodology. She is involved in clinical research and has published several original articles in peer-reviewed journals a The Lancet, Blood, Journal of Thrombosis and Haemostasis, Haematologica, Thrombosis and Haemostasis, British Journal of Haematology and Haemophilia. She is reviewer for several peer-reviewed journals and member of the Editorial Board of JTH. She is a member of several scientific societies (ISTH, WFH, ASH, EAHAD, SISET, AICE) and was a medical member of the Inhibitor Working Group of the European Hemophilia Consortium. She is co-chair of the ADVANCE Study Group. She has acted also as co-chair of the Scientific and Standardization Subcommittee of ISTH on FVIII, FIX and rare bleeding disorders. She has been involved as principal and co-investigator in several clinical trials, and she takes care of both children and adults with hemophilia and other congenital bleeding disorders with a specific scientific interest in novel therapies, prophylaxis, inhibitors, and chronic hepatitis C. As a patient with factor II deficiency, the diagnostic and treatment of rare bleeding disorders is a matter dear to my heart. My motivation to participate in the work of the ERIN committee is to improve both diagnostic and treatment for patients with rare bleeding disorders across Europe.
As a patient with factor II deficiency, the diagnostic and treatment of rare bleeding disorders is a matter dear to my heart. My motivation to participate in the work of the ERIN committee is to improve both diagnostic and treatment for patients with rare bleeding disorders across Europe. Economist and financial expert by profession, executive coach and trainer by passion and haemophilia advocate by every drop of my blood through my son (who has severe haemophilia A with inhibitors). Bringing a good decade of practical experience from the corporate insurance world, laser focus, growth mindset and resilience from my own experience, offering you anything I can just do, in hope that together we can make life more fulfilled for those impacted by bleeding disorders.
Economist and financial expert by profession, executive coach and trainer by passion and haemophilia advocate by every drop of my blood through my son (who has severe haemophilia A with inhibitors). Bringing a good decade of practical experience from the corporate insurance world, laser focus, growth mindset and resilience from my own experience, offering you anything I can just do, in hope that together we can make life more fulfilled for those impacted by bleeding disorders. Amy Owen-Wyard is a Registered Mental Health Nurse. With experience working with children, young people, their families and adults with severe and enduring mental health conditions. Amy was also involved in a service improvement to provide a holistic care approach for those in general hospitals to support both their mental and physical health, whilst sharing her expertise and knowledge in mental health with the wider multidisciplinary team.
Amy Owen-Wyard is a Registered Mental Health Nurse. With experience working with children, young people, their families and adults with severe and enduring mental health conditions. Amy was also involved in a service improvement to provide a holistic care approach for those in general hospitals to support both their mental and physical health, whilst sharing her expertise and knowledge in mental health with the wider multidisciplinary team.








